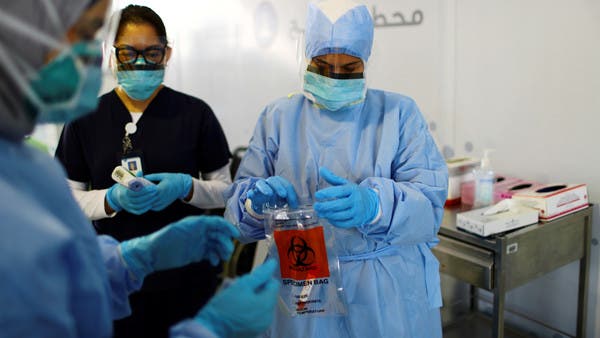
The coronavirus outbreak has led to governments world over fighting the pandemic to the best of their potential. From overwhelmed hospitals to people being laid off from their jobs to everything shutting down except for important businesses, COVID-19 has impacted lives in more ways than one. However, most countries remain determined, working day in and day out to develop a vaccine, which if successful can give hope for life to go back to normal. In an attempt to overcome the biggest challenge of the 21st century, the world’s first WHO-listed Phase III clinical trial of an inactivated vaccine kicked off in Abu Dhabi last month, with H.E. Sheikh Abdullah bin Mohammed Al Hamed, becoming the first volunteer to participate in the trial. Israeli Blogger Nas Daily, who is widely known for creating informative and realistic videos, recently paid a visit to the Gulf emirate and took his audience through the vaccine trial journey.
— ??????? ?? ???? (@ABZayed) August 8, 2020
He put together a video footage featuring a few of the total number of 15,000 volunteers who the vaccine is being tested on. He starts off with saying, “The world is racing. There are 150 Covid vaccines in development. Everyone is trying to find the cure.” Reporting live from the center in Abu Dhabi, Nuseir Yassin revealed that the COVID-19 vaccine from China has passed phase one and two, which means everyone who has taken it is safe from the virus. It is now in its final phase, giving more and more hope to people every day. If the vaccine works on them in phase three, it can be used for the whole world. Needless to say, this had us excited too! As the clip progresses, one volunteer shares, “When this works, we will go back to normal very soon.” Another adds, “Finally, for the first time in four months I feel helpful.” The ace video blogger then lauds the volunteers for their selfless act to humanity.
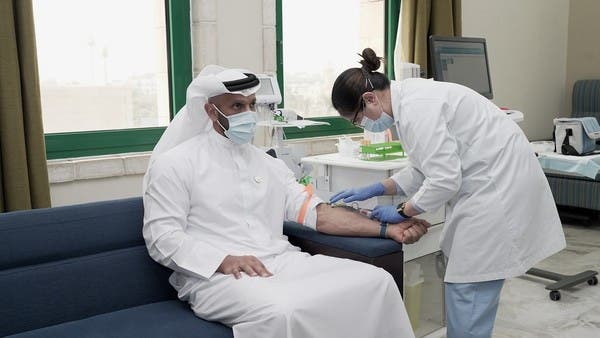
Registration for Volunteers
The world’s first phase three clinical trial of Covid-19 is being carried out under the #4Humanity campaign. The trials are being conducted in compliance with strict international guidelines stipulated by the World Health Organization (WHO) and the United States Food & Drug Administration (USFDA). One vaccine centre is the ADNEC, Zone 1,2,3 in Abu Dhabi and the other is Al Qarain Health Center in Sharjah. Volunteers are expected to visit a centre nearest to them, without prior appointment or registration. The centres are open seven days a week, from 8am until 8pm for on-the-spot registration, medical screening and vaccination.
Click on this link for complete information:
https://4humanity.ae/
The Volunteer Journey
Volunteers for the clinical trials must be over the age of 18 and in good health. They must register with official documents including ID/passport. They must either be a UAE resident or possess a valid residence permit during the trial period. Volunteers must sign a consent form before starting the trial. The first and second vaccination are 21-days apart from each other and Adverse Event Checks are conducted in between the two shots. After the 49th day, the volunteer will be contacted by their physician for a telephonic check-up once to twice monthly and physical check-up once every 3 months.
The duration of physical participation is approximately 2 months and the follow-up period might last up to 12 months. The date for all participants is kept confidential.
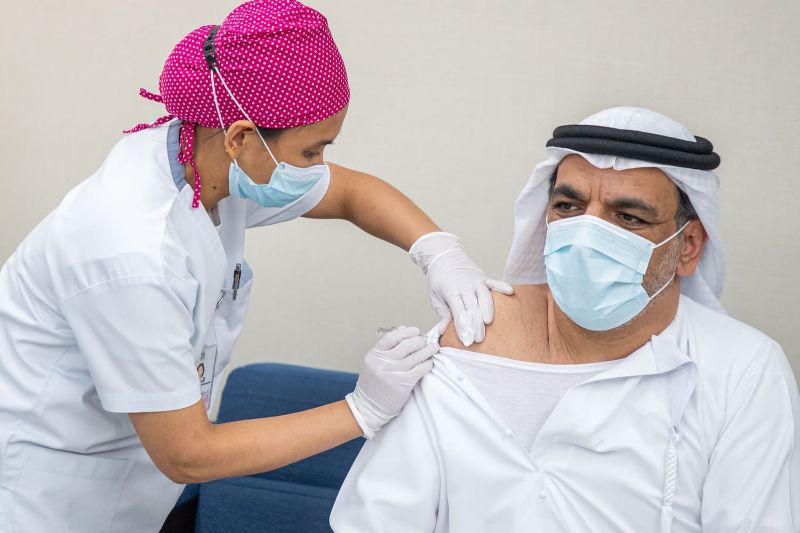
Who Can’t Volunteer?
Pregnant women are not permitted to volunteer. Moreover, those who have a history of SARS or MERS infection or who currently have a fever, dry cough, fatigue, nasal obstruction or runny nose within 14 days before vaccination are not allowed to volunteer either. This policy also applies to those who have a history of allergic reactions to medicines, convulsion or epilepsy, uncontrollable diabetes, any acute or chronic disease conditions like asthma, severe cardiovascular diseases, liver and kidney diseases, and malignant tumors. People currently receiving any immunotherapy or who have received blood within 3 months before the vaccination do not make the cut either.
Vaccine Trial Side-Effects
Some common reactions may occur when receiving this vaccine, including mild pain, redness, induration, and pruritus at the vaccination site. The volunteer may present mild systemic reactions such as fever, headache, fatigue, nausea, vomiting, diarrhea, cough, allergy, muscle pain, arthralgia, lethargy, etc. Usually these symptoms are mild and subside on their own. Patients with moderate to severe symptoms will be treated under the supervision of their treating physician and site principal investigator.
http://thebrownidentity.com/2020/08/09/dubai-design-district-to-host-d3-architecture-festival-2020/
Emergency Contact
Any symptom should be reported to the site principal investigator within 24 hours by visiting the healthcare clinic or calling on customer care at 02-8191112.